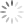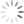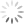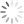by Elise Wachspress
Katie Harris came to the University of Chicago from Pittsburgh excited to apply her strong background in microbiology and immune signaling to advance the boundaries of science. She had found this post-doctoral opportunity—with eminent scientist Eugene Chang, MD—that would allow her to study how the microbiome contributes to inflammatory bowel disease, knowledge that someday might improve the lives of the 1.6 million people who suffer from this debilitating, often lifelong condition.
But she found UChicago offered an opportunity she never expected, one with impact much more immediate than “someday.” Never did she think she would be going into business.
It just so happened that a more advanced post-doctoral colleague in her lab, Joe Pierre, had just recognized some interesting things about a fungus named Candida albicans.
Candida, which grow well only in the human body, usually cause fairly solvable problems, like vaginal “yeast” infections. But Candida are shapeshifters. Usually oval (in the “yeast” mode), they can also convert to a hyphal form—a filament with a point at one end. And minor changes in its environment can cause rapid switching.
The problem is, hyphae can wreak havoc in the gut. They use their tips to break out of the human digestive system and into the blood stream. The result, for those with cancer or HIV or otherwise compromised immune systems, is a potentially deadly infection.
On the other hand, Candida in yeast form are valuable members of the gut community. They stimulate the gut lining, significantly reducing our susceptibility to inflammatory bowel disease, influenza infections, and other conditions. So eradicating Candida is not a winning strategy.
Another complicating factor is that fungal cells, unlike bacteria, are structurally similar to our own. This makes finding agents that target them without attacking human cells very difficult.
What Joe, now on faculty at the University of Tennessee, noticed was that there were certain peptides—smaller versions of proteins—that specifically prevent Candida from going from yeast to hyphal form. They are like fairy dust that could keep your basically good children always on their best behavior.
Joe’s next move was to pull together a team to make this discovery useful to the greatest number of people. He enlisted Katie and Dr. Chang, and they looked to the Polsky Center for Entrepreneurship and Innovation to help get the discovery out of the lab and into clinics and homes. The goal: develop a safe, effective product that would help prevent dangerous Candida infections in immuno-compromised patients. They started building a company: AVnovum, named for the novel AVpeptides they were creating to maximize the “fairy-dust” effect Joe had discovered.
The AVnovum team entered the Innovation Fund competition run by the Polsky Center. They knew that in order to be competitive they would need a strong business pitch as well as a good scientific idea, so they enlisted Myles Minter, a post-doctoral scientist at UChicago with a degree in intellectual property and an aptitude for business.
A large, experienced panel of business experts heard their pitch and awarded the team $150,000 to develop and test improved AVpeptides. Monica Vajani, a Booth student also trained as a biomedical engineer, enthusiastically joined the team to help. With Joe off to his faculty position in Tennessee and Myles accepting a position at William Blair, she and Katie became the “boots on the ground” to move this fledgling business forward.
Katie redoubled her efforts in synthesizing a variety of AVpeptides, about 30 so far, to see which worked the best to keep Candida in line. She first tests the peptides “in vitro”—in a petri dish where the Candida is growing by itself—and will eventually test in various animal models. She has already demonstrated the new compounds can improve effectiveness.
Meanwhile, Monica is collaborating closely with the Polsky team to put AVnovum on solid commercial footing. She developed a business plan, with the help of advisors at MATTER, who have extensive drug development experience. Once Katie identifies the best peptide candidates, they will concentrate on finding the most cost-effective ways to produce these in bulk, perhaps in the lab, or maybe via a biologic, using living microorganisms, plants, or animal cells to manufacture the compounds. Next will come preclinical toxicology tests, clinical trials, etc.: a long road, but perhaps a profitable one. Candida poses a $3.5 billion burden to the health care system, and those with Candida bloodstream infections have mortality rates as high as 30 percent. There are currently no safe and specific treatments for this subset of immunocompromised patients.
The AVnovum team hopes to change that fact soon. The peptides can be given orally and are so specific to defanging Candida that demonstrating safety is likely a slam-dunk. For those at highest risk—those on high-dose corticosteroids or about to receive an organ transplant or being treated for cancer—the AVpeptides will likely be used prophylactically, to prevent infections in the first place.
Katie has surprised herself with her own involvement in this business venture. “Entrepreneurial science was something I never had an interest in,” she says. “I got involved with biological research because I find the natural world absolutely fascinating.
“But I am thrilled to know that my work can actually improve lives. If this treatment ends up available to patients, my work in the lab would contribute to preventing fungal infections in a safe and effective way. I still love performing research in basic biological concepts, but I now have a greater appreciation for how that research can be used to improve human health.”
As sources of government and corporate funding flow and (mostly) ebb, capturing the monetary value of scientific discovery may be essential to advancing the boundaries of knowledge. Good science is expensive, but it is the surest road to progress.
That is why the growing partnership between the University of Chicago Medicine and the Polsky Center for Entrepreneurship and Innovation may be one of the most critical facets of the Duchossois Family Institute.
Photo above (cropped): Candida albicans in both yeast and hyphal forms. The rough-surfaced cell at the center is a neutrophil, one of the immune system’s “first-responders” to infection or environmental toxins. (Kernien, John F.; Johnson, Chad J.; Nett, Jeniel E., CC BY 4.0)
Elise Wachspress is a senior communications strategist for the University of Chicago Medicine & Biological Sciences Development office