Jan 3, 2020 | Cancer, Immunology, Microbiome, News Roundup
A selection of health news from the University of Chicago and around the globe curated just for you.
Microbes living in the tons of plastics in the oceans
Jessica Mark Welch and colleagues at UChicago’s Marine Biological Laboratory at Woods Hole, MA, aim their new microscopy technique at microplastic samples from the ocean to characterize the extensive biofilms on ocean plastic. (Agro Ecology Innovations)
Staph vaccines likely to work better if administered earlier
Research by former UChicago faculty member Julian Bubeck-Wardenburg, now at Washington University in St. Louis, suggests that vaccinating infants before their first encounter staph, either just after birth or via their mothers during pregnancy, would likely generate a stronger immune response. (Futurity)
Microbial inequity
To the host of ways people experience inequity, add the microbiome: A University of Maine scientist argues that access to fibrous foods, parks, good air quality, other infrastructure affect the development of a healthy microbiome. (The Conversation)
UChicago faculty lauded yet again
Thomas Gajewski, MD, PhD, Abbvie Foundation Professor of Cancer Immunotherapy, is honored with the 2019 Award for Immuno-Oncology from the European Society for Medical Oncology (Healio)
Gut neurons are anti-salmonella warriors
Research from Harvard Medical School have found that nerves in the gut not only regulate the cellular gates that admit microorganisms, but actively boost the number of protective microbes there. (ScienceBlog)

Dec 5, 2019 | Cancer, Immunology, Microbiome
A selection of health news from the University of Chicago and around the globe curated just for you.
The community in your mouth
Marine Biological Laboratory and DFI researcher Jessica Mark Welch, PhD, shares her research into oral microbiome, and what we can learn about healthy and diseased states from microbes’ relationships and interactions. (Smithsonian Magazine)
Common drugs linked with microbiome alterations
Patients who used drugs from 18 common drug classifications—including proton pump inhibitors, metformin, antibiotics, laxatives, and others—showed extensive changes in their gut microbiomes. (MedPage Today)
DFI researcher Tatyana Golovkina, PhD, named AAAS fellow
UChicago professors Melina Hale, PhD, and Margaret Beale Spencer, PhD’76, were also honored by the American Association for the Advancement of the Sciences—the world’s largest general scientific society. (UChicago News)
Urine microbiome predicts cancer immunotherapy response
UChicago physician-scientist Randy Sweis, MD, demonstrated that the presence of certain strains of bacteria could tell us which bladder cancer patients would face recurrence after immunotherapy treatment. (Healio)
Diets high in fiber and yogurt cut lung cancer risk
By collecting data from several lung cancer studies involving more than 1.4 million adults from the US, Europe, and Asia, researchers showed that high-fiber, high-yogurt diets reduced lung cancer risk by one third. (Futurity)

Nov 5, 2019 | Cancer, Immunology
by Paulette Krishack
Post-doctoral fellow in the University of Chicago’s Department of Medicine
Breast cancer is one of the most commonly diagnosed cancers in women. Approximately one in eight will develop the disease in their lifetime, and over 40,000 in the U.S. will die from it this year. While genetics plays a role, 85 percent of women diagnosed with breast cancer have no family history, and effective prevention and treatment depends on identifying the other factors involved.
Clinicians break down breast cancers into at least five types, based on the molecular features present in the tumor cell. Several types are known to show hormone “receptors” (most commonly estrogen, progesterone, and a protein called HER-2) which offer targets for treatment.
But in one type, triple negative breast cancer (TNBC), tumor cells lack the hormone receptors and HER-2 protein known to fuel breast cancer. Finding a way to specifically target TNBC, a particularly aggressive form of cancer, is critical.
Obesity is known as a risk factor for breast cancer incidence and progression. Because obesity can cause chronic, systemic inflammation, Lev Becker, PhD, Marsha Rosner, PhD, and Payal Tiwari, PhD, were interested in finding out how the immune system might be involved in TNBC.
Becker has dedicated his laboratory and career to studying macrophages, the “big eaters” of the immune system, while Rosner specializes in cancer therapeutics. Although macrophages are powerful fighters of viruses and bacteria, they can also be called in to engulf the saturated fatty acids found in many high-fat foods, causing high-fat induced inflammation in the tissues they invade. Becker, Rosner, and Tiwari wanted to see if these obesity-associated macrophages were involved in TNBC.
Using a mouse model of TNBC, they found that the most common macrophages in the breast fat tissue of obese mice were a type specifically activated by a high-fat diet. These macrophages produced a pro-inflammatory protein, a cytokine called IL-6, which created a nurturing environment for TNBC tumors to grow. The team also found that switching the obese mice to a low-fat diet slowed tumor development. Studies also showed human tumor tissue with similar results: there were fewer macrophages present in tissues from lean patients than obese ones.
These studies suggest that immunotherapy—treatments targeting the immune response that is activated by macrophages—may offer innovative care for patients with TNBC, especially if they are obese. They may also offer potential strategies for treating other diseases linked to obesity, particularly those that involve macrophages, such as cardiovascular disease.
Most importantly, this research demonstrates the value of diet in maintaining health. A third of American adults are obese and another third overweight. Now that we know the toll a high-fat diet exerts on molecules in our body, we can embrace the mission of the Duchossois Family Institute: Use the knowledge we develop to improve our health and all those with whom we share the planet.

Sep 16, 2019 | Cancer, Immunology, Microbiome
by Elise Wachspress
In the world of cancer research, Thomas Gajewski, MD, PhD, is a rock star. And not just because he plays lead guitar for The Checkpoints, an all-cancer-researcher band that includes his longtime colleague Jim Allison, last year’s Nobel Prize winner for physiology or medicine (and professor at the MD Anderson Cancer Center) on harmonica.
Named a Giant of Cancer Care in 2017, Gajewski has, for more than two decades, worked in cancer immunotherapy, a field only recently recognized as the most promising for actual cancer cures—a word that makes most clinicians nervously back away.
Gajewski has dedicated his career to figuring out how to help patients’ immune systems fight cancer from within. Much of his work has focused on checkpoint inhibitors, proteins which our bodies use to keep immune responses in check, so we don’t attack beneficial bacteria or our own cells. But checkpoint inhibitors can also keep our immune system from killing cancer cells (and thus the name chosen for Gajewski’s band).
Over the past century, the idea of fighting cancers with immunotherapies has fallen in and out of favor multiple times. But oncologists are now watching the field—and Gajewski’s work—with excitement. Recent research shows that, in metastatic melanoma and many other solid cancers, immunotherapies cause tumors to regress or even disappear in 30-50% of patients. These treatments are often significantly less toxic than surgery, radiation, or traditional chemotherapy. And by training the body to recognize and kill cancer cells, immunotherapies offer an opportunity for truly durable results, offering patients a chance at living long, healthy, cancer-free lives.
But Gajewski, the AbbVie Foundation Professor of Cancer Immunotherapy, is focused past these success stories. What drives his curiosity and his passion is understanding why immunotherapies fail. And one of his most remarkable findings is that the bacteria patients carry in their guts—their microbiome—can strongly influence whether or not cancer immunotherapy works.
As with most scientific breakthroughs, this one came through a combination of dogged dedication, careful observation, and wonder. When Gajewski and his team implanted mice with melanoma tumors, they noticed immunotherapy response differed depending upon the commercial source of the animals themselves; one supplier’s mice fared much better, even though they were identical strains. The team finally found that the mice were delivered to the lab carrying a certain type of gut bacteria responded much better to the therapy.
Even more tantalizing: when the two sets of mice were housed together, outcomes improved for the mice from the other supplier. Sharing these healthy gut bacteria was key to therapeutic success.
Gajewski and team are now working to identify bacteria found in humans that can produce these effects in mice. The team will use a system in which germ-free mice are colonized with microbial material from patients, implanted with tumors, and then treated with immunotherapy drugs. The goal is to figure out the exact mechanism of how bacteria promote or restrict anti-tumor immunity—knowledge that will provide a foundation for new therapeutics. By developing computational algorithms which integrate genomic sequencing of the microbiota, the tumor oncogenes, and germline polymorphisms, they aim to get a comprehensive view of immunotherapy success versus resistance in each individual patient.
The goal is a diagnostic approach that will help clinicians—like Gajewski himself—decide the most effective way to use immunotherapy for each patient and tumor, perhaps by simply introducing new types of bacteria to a patient’s gut. While we commonly think of probiotics as a way to improve digestion or alleviate allergies, we may one day find an avenue to use much more sophisticated, designer probiotics to treat some cancers.
Gajewski recently received a promise of nearly half a million dollars from the Melanoma Research Alliance to advance this research. However, securing this award requires him to match the grant with similar funding from other sources.
For anyone ready to invest in much more effective, less debilitating treatments—perhaps even cures—for cancer, Gajewski has a strategy ready for prime time, a track record of unusual research success, and a group of musicians ready to sing about it.
Elise Wachspress is a senior communications strategist for the University of Chicago Medicine & Biological Sciences Development office
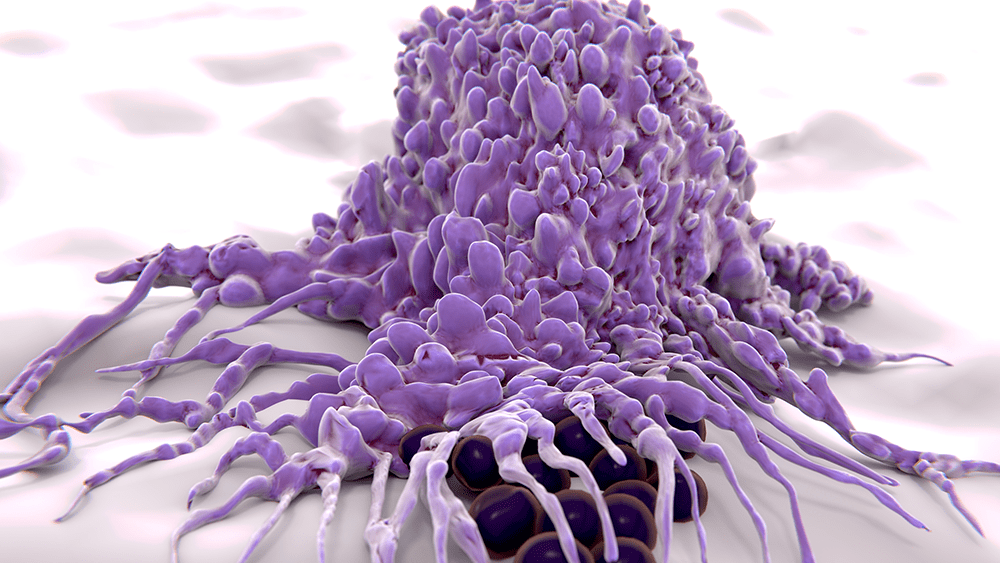
May 14, 2019 | Cancer
by Tim McGovern
Writer and editor based in Chicago
Remember The Blob? Even if you’ve never seen this 1958 B-movie classic, pop culture has likely introduced you to the concept: a shapeshifting mass that extends to swallow up and dissolve its enemies. This swallow and dissolve approach—on a microscopic level—is how white blood cells deal with infections. As Lev Becker, who specializes in these cells, explains: “These macrophages (their very name means ‘big eaters’) and neutrophils (the ‘first-responder’ white blood cells) are in every tissue in your body, and they play a fundamental role in maintaining your health, protecting you against infections from viruses or bacteria.”
Unlike the Blob and its strategy of indiscriminate destruction, white blood cells do a pretty good job of distinguishing the “good guys”—healthy human tissue—from the “bad guys”—viruses and bacteria. But sometimes they get it wrong. Becker goes on: “If they’re chronically incorrect—for instance, over-inflamed in the context of diabetes and heart disease, or under-inflamed in the context of cancer—long-term illnesses can result. And although we are looking at a variety of diseases, what unifies our lab is the philosophy and approach that one can either activate or inhibit the innate immune system to treat human disease.”
To understand how immune cell activity plays out in human disease, Becker and his team use an array of leading-edge techniques. “We always start with people,” he says, “and ask ‘What is different in the cells of people with and without a given disease?’ Then we work to find mouse models and cell systems that replicate that dysfunction.” This approach sets his lab apart from a more “traditional” approach: start with a molecule and learn everything about it in the cellular context, then—if it seems the molecule might play a role in disease—develop a mouse model, and finally, if the mouse results are promising, figuring out a way to ascertain the molecular activity in humans.
In Becker’s lab, “Every project has a human, mouse, and cell component to it.” It takes a novel assortment of tools to work in this way. In addition to the core biochemistry the team employs to parse the molecular pathways, they have developed new assays using cutting-edge computational techniques, with the goal of understanding the entire range of protein activity in a given cell. The capstone to all this molecular research is the lab’s ability to test their hypotheses not only in petri dishes, but in mice and human patients.
This ability to take research from human to molecule to human was always part of Becker’s plan: “My initial thought going into basic science versus medical science is that to impact health you have to take it one step further. Research scientists always hope that one day their findings will lead to treatments, but our focus on the translation part of research is especially important to us. We want to create medical interventions and develop therapeutics. That’s where we’re trying to go: start with humans, go back, find a molecule, take it to mice, and then launch a company, get funding, and move forward with actually developing and testing a therapy.”
A couple of years ago, leveraging the expertise and resources of the University’s Polsky Center for Innovation and Entrepreneurship, Becker launched Onchilles Pharma Inc. With seed funding from the George Shultz Innovation Fund, Onchilles is working to bring Becker’s research back to people, in the form of cancer immunotherapies which are more powerful, more precise, and better tolerated than current treatments.

Apr 30, 2019 | Cancer, Microbiome, News Roundup
A selection of health news from the University of Chicago and around the globe curated just for you.
Providing a community of support for young women
The region’s first pediatric lupus clinic opens at Comer Children’s Hospital thanks to the generosity of the Handley family and Lakeshore Recycling Systems. The clinic aims to help adolescents manage the physical and psychosocial challenges of living with lupus. Melissa Tesher featured. (Give to Medicine)
Drug delivery technique makes cancer immunotherapy more effective
Researchers in the Institute for Molecular Engineering at the UChicago have developed a new way to target immunotherapies and deliver them directly to tumors, helping to both reduce side effects and make the therapies more effective in treating cancer. Jeffery Hubbell and Melody Swartz featured. (UChicago Medicine)
Your sexual partners can change your microbiome, a new study in mice finds
People’s sexual partners could impact both their gut microbiome and their immune system, according to a new study from the University of Colorado, Denver. (STAT)
Gut microbiome directs immune system to fight cancer
Researchers identified a cocktail of 11 bacterial strains that activated the immune system and slowed the growth of melanoma in mice. Thomas Gajewski quoted. (Drug Target Review)
The Microbiome Center continues its focus on the broader world of microbes
Co-director Cathy Pfister, PhD, says the resources and research infrastructure being developed by the DFI are a tremendous boon for all researchers studying the microbiome at UChicago. The DFI’s emphasis on human health also allows the Microbiome Center to focus its efforts across many environments. (UChicago Medicine)