Quanteon
David and Etta Jonas Center for Cellular TherapyHome » Benchtop Analyzers » David and Etta Jonas Center for Cellular Therapy
Overview
The CAT Facility manages the David and Etta Jonas Center for Cellular Therapy flow cytometry equipment located in the S301 satellite facility in the Billings Hospital. Any user can access the instrument, however, Jonas Center members have priority on scheduling every day from 1PM to 5PM. Walk-ins by non-Jonas Center members are allowed when the instrument is not booked during that period.
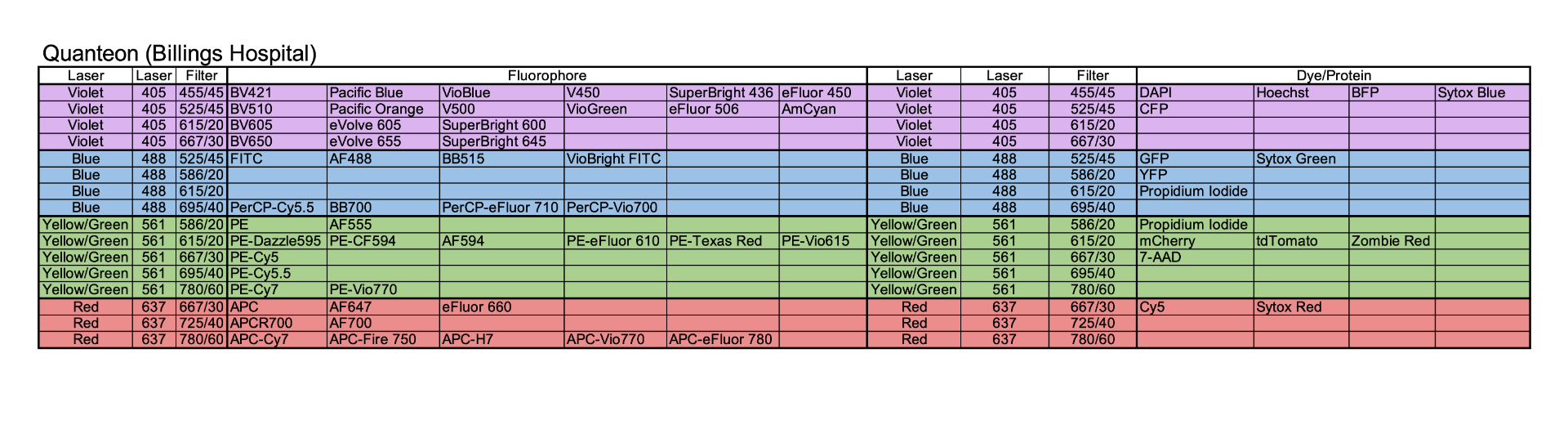
Model: NovoCyte Quanteon
Manufacturer: Agilent
Located in Billings Hospital S301
Additional Features:
- NovoSampler Q compatible with 40 tube rack, and 24- 48- 96, and 384-well plate
- Absolute cell count
Resources
Software Tutorial
Troubleshooting
Coming Soon
Startup - First user in the day
- Check on the Fluidics tank: everything full, waste empty. NOTE - remove the tanks from the fluidic station before filling them up, spills may damage the electronic balance at the bottom of the unit.
- Power on the instrument - it will take ~6 minutes before the instrument is ready to go.
- Open your Windows session and the software.
- Run the QC Test protocol, from the instrument menu (see below for instructions).
If the instrument has already been started by another user, you do not have to repeat these steps.
How and when should I run the QC Test
Run the QC Test whenever you turn the instrument ON. The test should take ~6 minutes.
1 - Prepare the beads: vortex the tube, add 1 drop to 0.5ml of diH2O
2 - Make sure the correct plate or rack is selected
3 - From the Instrument menu, check QC Test
3 (optional) - save the file when prompted
4 - Check the bead lot and location of the bead sample on the plate, press Run
5 - The software runs 3 tests: Electronic Noise, Optical Noise, and the QC Test
6 - Everything should pass. Contact the CAT Facility if anything fails.
How do I use the Compensation Wizard / The Compensation Wizard is acting funny
Below is a training video on the NovoExpress Auto Compensation tool. There's a few hiccups with the tool: 1 - the software decides the location and order of the controls on the plate, which may not fit what you decided when you loaded your plate. We suggest adding your compensation controls in tubes so you can re-arrange the order on the rack. 2 - Once the compensation matrix is calculated, you won't be allowed to run samples on any other platform then the one that you used for your controls. One way around that is to create an experiment template containing your compensation matrix. See video for details.
No data is showing
- It is possible to open multiple instances of the NovoExpress software, having one of them connected to the instrument, while looking at previously saved experiments. So make sure that you are looking at the one that should be connecting to the instrument.
- Reboot the computer re-open the session and software.
- If that fails, power cycle both the instrument and the computer. This may trigger the instrument to do its 20 minutes clean-up protocol, followed by the 6 minutes startup. Sorry.
'Collision of SIP' error message
1 - you have not selected the proper plate or tube rack in the Plate Manage Window, make the appropriate modification.
2 - you are using a whole new type of plate on the instrument and it has not been calibrated for it (contact the staff for assistance)
3 - the NovoSampler Q needs to be recalibrated (go to Instrument -> Calibrate)
End of session
- From the Template menu, open the 3-tubes clean SIP protocol, and load tubes of 3ml of NovoClean, NovoRinse and water in position A1, A2, and A3 respectively on the rack. Let the instrument complete the protocol
- Close the software and close your windows session
If you are the user in the day, turn off the instrument by pushing the power button. This will trigger a 20 minutes flow cell cleaning protocol. You are not required to stay during the protocol.
Data will not upload to the server
- The data should upload automatically every 30 minutes, so be sure that you gave enough time for the transfer to occur.
- Data should b exported to D:/NovoExpress Data. Make sure that you did not export your files in another location - either somewhere else on the disk, or a sub-folder of NovoExpress data.
- In some case communication gets lost, contact the staff and we will alert our IT manager.