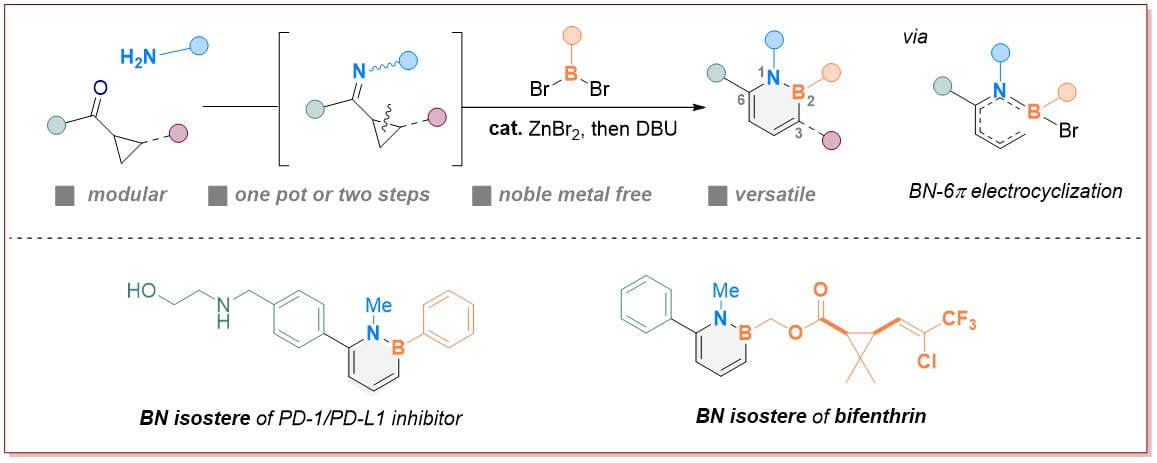
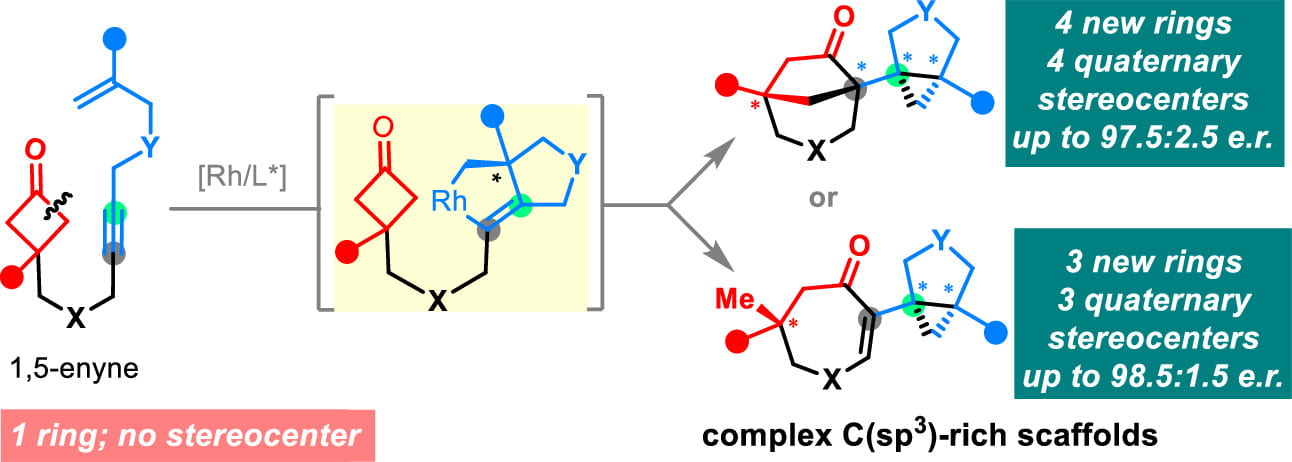
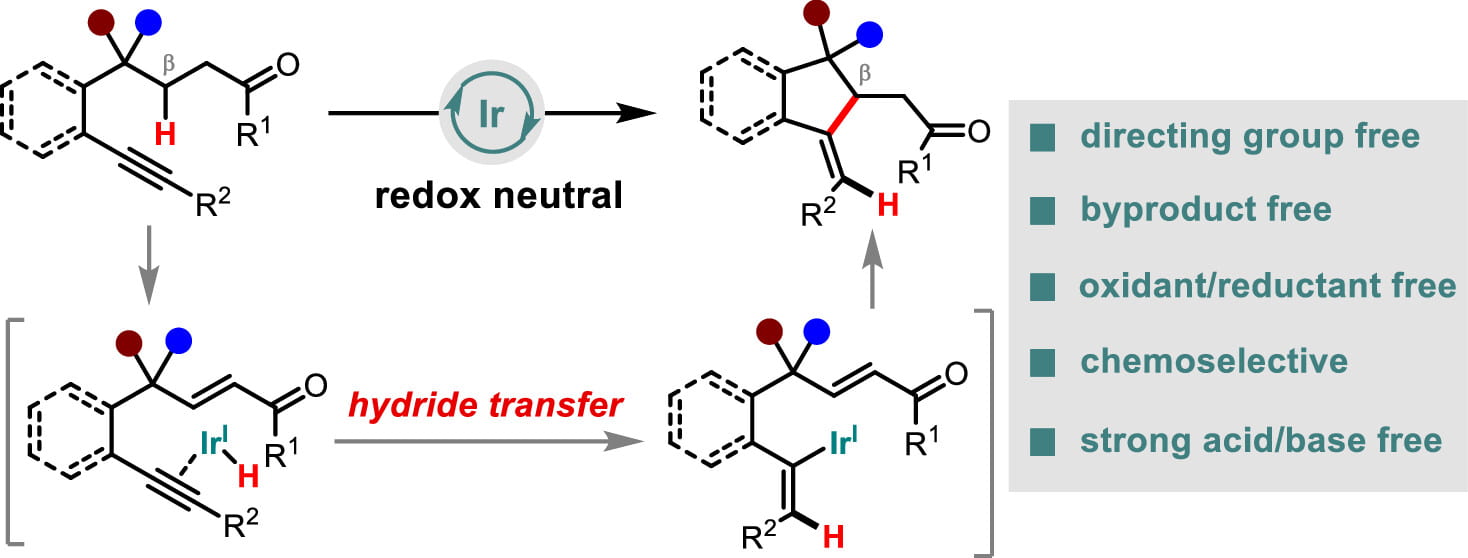
Research Directions
Our research interests is to harness the power of transition-metal catalysts to address the challenges in the arena of chemoselectivity and multi-step synthesis. Our research philosophy is guided by three questions:
1) What would be the most efficient way to introduce functional groups into (small or large) organic molecules?
2) What would be the most efficient method to synthesize various molecular skeletons (bridged or fused rings) with high complexity?
3) What would be the most efficient strategy to access mutiple structurally complicated and biologically important natural products and their analogues in a rapid fashion?

NEWS
Congrats! Rui, Miao and Zining!
Three talented graduate students in our lab, Rui, Miao, and Zining, have been awarded the Elizabeth R. Norton Prize for Excellence in Research in Chemistry, the Frances E. Knock Scholarship, and the Everett E. Gilbert Memorial Prize for the Best Third-Year...
Congrats, Shinyoung
Congratulations to Dr. Shinyoung Choi for defending her Ph.D.! She will move to SK Innovation in South Korea! We wish her all the best in her new page of life!