It’s time to get back to the Bad Data series. I’ll use a data set I had to troubleshoot for one of my users to illustrate the importance of cleaning your data. We mentioned FlowAI and FlowClean in the first installment of this series, and this post will hopefully showcase how useful they can be.
My user presented me with the dot plot below where can see a decent amount of very negative events displayed on the PacBlue axis.

Pseudocolor plot of EYFP and CD4 PacBlue, where the events have been gated on FSCxSSC only.

NxN plot reveals no obvious compensation issue with the data.

Pseudocolor plot of Time x PacBlue. Weirdness includes inconsistent number of events acquired over time, and downward spikes in fluorescence on the PacBlue parameter.
Here, I used FlowAI to clean up the data. There are several other algorithms that will perform a similar task, and I can’t claim to be able to explain the specifics of how each of them perform. But roughly speaking, they will find the data that doesn’t look right and remove it from the analysis. Once the data is cleaned up, the final analysis will look much better.

FlowAI removed the portion of the data that didn’t look like the rest

All is good in the world
It will be important to understand what the algorithm is doing to your data in order to figure out to what degree you can trust in the remaining data. Publications on your favorite tools are available on PubMed and elsewhere.
One way to mitigate your need for these solutions is to work on your sample preparation – optimize your single cell suspension, filter your samples, etc. If you need tips and tricks, check out our Flow Basics 2.0 class.
For this presentation, I ran FlowAI in FCS Express. See below for a very quick overview of how it was done. You will find a similar toolbox on other analysis platforms available at the CAT Facility such as FlowJo and OMIQ.



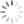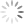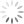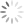
You help me out with Flow Cytometry Data Analysis.
Thank you
Send us your data by email and we’ll have a look. If we think it will require a sensible amount of our time, we’ll provide you with a quote to perform the work.